We serve 3-(Benzoylthio)-2-methylpropanoic acid CAS:74431-50-8 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
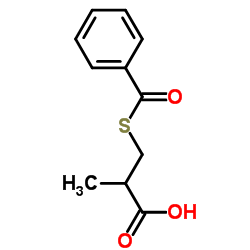
Contact us for information like 3-(Benzoylthio)-2-methylpropanoic acid chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,3-benzoylsulfanyl-2-methylpropanoic acid physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,3-benzoylsulfanyl-2-methylpropanoic acid Use and application,3-benzoylsulfanyl-2-methylpropanoic acid technical grade,usp/ep/jp grade.
Related News: The company’s short-term plan is to move from a purely chemical intermediate manufacturer to a formula active material supplier, and its long-term goal is to become a solution provider in the selected segment. In the future, this business is expected to become a new growth point for the company’s profit.2-phenylethyl isocyanate manufacturer Onconova Therapeutics, Inc. and Inceptua Medicines Access (a business unit of the Inceptua Group) recently announced they have entered into a collaboration to make available intravenous rigosertib via a Pre-approval Access Program in selected countries around the world.(R)-BoroLeu-(+)-Pinanediol-HCl supplier Onconova Therapeutics, Inc. and Inceptua Medicines Access (a business unit of the Inceptua Group) recently announced they have entered into a collaboration to make available intravenous rigosertib via a Pre-approval Access Program in selected countries around the world.(2R)-Methyl 2-acetamido-3-chloro-3-hydroxypropanoate vendor Onconova Therapeutics, Inc. and Inceptua Medicines Access (a business unit of the Inceptua Group) recently announced they have entered into a collaboration to make available intravenous rigosertib via a Pre-approval Access Program in selected countries around the world.“With early evidence of clinical activity for our off-the-shelf, iPSC-derived NK cell programs, we are excited to lead in bringing next-generation CAR T-cell therapies to patients and plan to submit an IND for FT819 in the first half of 2020.”

