We serve 2-Bromo-3-fluorophenol CAS:443-81-2 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
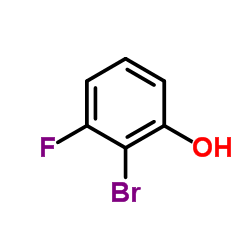
Contact us for information like 2-Bromo-3-fluorophenol chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,3-Fluoro-2-bromophenol physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,2-Bromo-3-fluorophenol Use and application,3-Fluoro-2-bromophenol technical grade,usp/ep/jp grade.
Related News: The market for pharmaceutical intermediates is vast and is expected to usher in new growth. Compared with the pesticide intermediate business, the market space for pharmaceutical intermediates is larger and will be the focus of the company’s future development.4-Bromo-2-nitro-6-(trifluoromethyl)aniline manufacturer Most foreign drugmakers offered Beijing the lowest prices globally in a recent round of negotiations in order to get some of their new products included in national insurance program, a move that will help them gain access to more patients in less-affluent cities, officials said on Thursday.Bis(2-chloroethyl)amine hydrochloride supplier Most foreign drugmakers offered Beijing the lowest prices globally in a recent round of negotiations in order to get some of their new products included in national insurance program, a move that will help them gain access to more patients in less-affluent cities, officials said on Thursday.Siloxanes and Silicones, Me 3,3,3-trifluoropropyl, Me vinyl, hydroxy-terminated vendor In order to find answers to these questions, our staff in the development department set about conducting a series of experiments.From the perspective of the corresponding formulation manufacturer, the drug substance needs to meet the requirements of impurities and stability. The production base must pass the international quality system certifications such as cGMP and EuGMP. At the same time, the drug substance company must have sufficient capacity.

