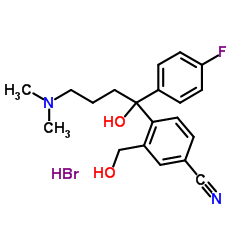
We serve 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile hydrobromide CAS:103146-26-5 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
Contact us for information like 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile hydrobromide chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,Benzonitrile, 4-[4-(dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl]-3-(hydroxymethyl)-, hydrobromide physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile hydrobromide Use and application,4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile hydrobromide technical grade,usp/ep/jp grade.
Related News: Taking Minuo Huawei as an example, in 2016, the capacity utilization rate and production-sales ratio of its multiple drug substance varieties reached more than 90%. Therefore, domestic API companies have expanded their production capacity in recent years to meet the growing demand for APIs.N,O-Dimethylhydroxylamine hydrochloride manufacturer Taking Minuo Huawei as an example, in 2016, the capacity utilization rate and production-sales ratio of its multiple drug substance varieties reached more than 90%. Therefore, domestic API companies have expanded their production capacity in recent years to meet the growing demand for APIs.(R)-4,5,6,7-Tetrahydro-benzothiazole-2,6-diamine supplier Taking Minuo Huawei as an example, in 2016, the capacity utilization rate and production-sales ratio of its multiple drug substance varieties reached more than 90%. Therefore, domestic API companies have expanded their production capacity in recent years to meet the growing demand for APIs.Divinyltetramethyldisiloxane vendor Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).

