We serve 2-bromo-1,1,1,2,3,3,3-heptafluoropropane CAS:422-77-5 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
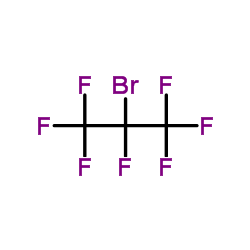
Contact us for information like 2-bromo-1,1,1,2,3,3,3-heptafluoropropane chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,2-bromo-heptafluoro-propane physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,2-bromo-1,1,1,2,3,3,3-heptafluoropropane Use and application,2-bromo-1,1,1,2,3,3,3-heptafluoropropane technical grade,usp/ep/jp grade.
Related News: Massachusetts state health officials, in conjunction with Massport, local health departments, and other medical partners, have responded to prevent the spread of the virus.4′-((5-butyl-N-(3-phenylpropyl)picolinamido)methyl)-[1,1′-biphenyl]-2-carboxylic acid manufacturer Massachusetts state health officials, in conjunction with Massport, local health departments, and other medical partners, have responded to prevent the spread of the virus.4-(2-benzofuran-4-yl-5-oxo-oxazol-4-ylidenemethyl)-N-phenyl-piperazine-1-carboxamidine hydrochloride supplier Massachusetts state health officials, in conjunction with Massport, local health departments, and other medical partners, have responded to prevent the spread of the virus.N-{(R)-4-[1-(3-Cyclopentyloxy-4-methoxyphenyl)-2-(pyridin-4-yl)ethyl]phenylacetyl}benzenesulfonamide vendor From the perspective of the corresponding formulation manufacturer, the drug substance needs to meet the requirements of impurities and stability. The production base must pass the international quality system certifications such as cGMP and EuGMP. At the same time, the drug substance company must have sufficient capacity.From the perspective of the corresponding formulation manufacturer, the drug substance needs to meet the requirements of impurities and stability. The production base must pass the international quality system certifications such as cGMP and EuGMP. At the same time, the drug substance company must have sufficient capacity.

