We serve Chemical Name:5-Amino-3-hydroxyvaleric acid CAS:63316-28-9 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
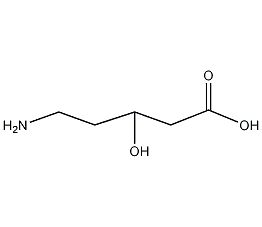
Chemical Name:5-Amino-3-hydroxyvaleric acid
CAS.NO:63316-28-9
Synonyms:5-Amino-3-hydroxyvaleric acid
Molecular Formula:C5H11NO3
Molecular Weight:133
HS Code:
Physical and Chemical Properties:
Melting point:N/A
Boiling point:N/A
Density:N/A
Index of Refraction:
PSA:
Exact Mass:
LogP:
Material Safety Information (Applicable for Hazard Chemicals)
RIDADR:
Packing Group:
Contact us for information like 5-Amino-3-hydroxyvaleric acid chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,5-Amino-3-hydroxyvaleric acid physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,5-Amino-3-hydroxyvaleric acid Use and application,5-Amino-3-hydroxyvaleric acid technical grade,usp/ep/jp grade.
Related News: The review considered two cases of AML in patients treated with an investigational medicine, bb1111, in a clinical trial for sickle cell disease. Although there have been no reports of AML with Zynteglo, both medicines use the same viral vector and there was a concern that the vector may be implicated in the development of the cancer (insertional oncogenesis). 2,5-Dimethyl-4-(triphenyl-silyl)pyridin-1-oxid manufacturers Inspired by the spirit of Start with Integrity, Succeed through Action,” Innovent’s mission is to develop, manufacture and commercialize high-quality biopharmaceutical products that are affordable to ordinary people.
Established in 2011, Innovent is committed to developing, manufacturing and commercializing high-quality innovative medicines for the treatment of cancer, autoimmune, metabolic and other major diseases. On October 31, 2018, Innovent was listed on the Main Board of the Stock Exchange of Hong Kong Limited with the stock code: 01801.HK.
Since its inception, Innovent has developed a fully integrated multi-functional platform which includes R&D, CMC (Chemistry, Manufacturing, and Controls), clinical development and commercialization capabilities.
Leveraging the platform, the company has built a robust pipeline of 25 valuable assets in the fields of cancer, metabolic, autoimmune disease and other major therapeutic areas, with 5 products – TYVYT® (sintilimab injection), BYVASDA® (bevacizumab biosimilar injection), SULINNO® (adalimumab biosimilar injection), HALPRYZA® (rituximab biosimilar injection) and Pemazyre® (pemigatinib oral inhibitor) – officially approved for marketing, 1 asset’s NDA under NMPA review, sintilimab’s Biologics License Application (BLA) acceptance in the U.S., 5 assets in Phase 3 or pivotal clinical trials, and an additional 14 molecules in clinical studies.
Innovent has built an international team with advanced talent in high-end biological drug development and commercialization, including many global experts. The company has also entered into strategic collaborations with Eli Lilly and Company, Adimab, Incyte, MD Anderson Cancer Center, Hanmi and other international partners.
Innovent strives to work with many collaborators to help advance China’s biopharmaceutical industry, improve drug availability and enhance the quality of the patients’ lives. For more information, please visit Ac-Ser(BZL)-Asn-Ala-Ala-Val-Asp(OBZL)-Thr(BZL)-Ser(BZL)-Ser(BZL)-Glu(OBZL)-Ile-Thr(BZL)-Thr(BZL)-Lys(Z) suppliers Mene Pangalos, executive vice president, BioPharmaceuticals R&D, said: “The data being presented at EULAR add to the growing body of evidence for anifrolumab that demonstrate a compelling clinical profile with the potential to address significant unmet medical needs in this debilitating disease. β-hydroxydehydroabietic acid vendor & factory.
Established in 2011, Innovent is committed to developing, manufacturing and commercializing high-quality innovative medicines for the treatment of cancer, autoimmune, metabolic and other major diseases. On October 31, 2018, Innovent was listed on the Main Board of the Stock Exchange of Hong Kong Limited with the stock code: 01801.HK.
Since its inception, Innovent has developed a fully integrated multi-functional platform which includes R&D, CMC (Chemistry, Manufacturing, and Controls), clinical development and commercialization capabilities.
Leveraging the platform, the company has built a robust pipeline of 25 valuable assets in the fields of cancer, metabolic, autoimmune disease and other major therapeutic areas, with 5 products – TYVYT® (sintilimab injection), BYVASDA® (bevacizumab biosimilar injection), SULINNO® (adalimumab biosimilar injection), HALPRYZA® (rituximab biosimilar injection) and Pemazyre® (pemigatinib oral inhibitor) – officially approved for marketing, 1 asset’s NDA under NMPA review, sintilimab’s Biologics License Application (BLA) acceptance in the U.S., 5 assets in Phase 3 or pivotal clinical trials, and an additional 14 molecules in clinical studies.
Innovent has built an international team with advanced talent in high-end biological drug development and commercialization, including many global experts. The company has also entered into strategic collaborations with Eli Lilly and Company, Adimab, Incyte, MD Anderson Cancer Center, Hanmi and other international partners.
Innovent strives to work with many collaborators to help advance China’s biopharmaceutical industry, improve drug availability and enhance the quality of the patients’ lives. For more information, please visit Ac-Ser(BZL)-Asn-Ala-Ala-Val-Asp(OBZL)-Thr(BZL)-Ser(BZL)-Ser(BZL)-Glu(OBZL)-Ile-Thr(BZL)-Thr(BZL)-Lys(Z) suppliers Mene Pangalos, executive vice president, BioPharmaceuticals R&D, said: “The data being presented at EULAR add to the growing body of evidence for anifrolumab that demonstrate a compelling clinical profile with the potential to address significant unmet medical needs in this debilitating disease. β-hydroxydehydroabietic acid vendor & factory.

