We serve Chemical Name:Iomeprol Impurity 2 CAS:104517-95-5 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
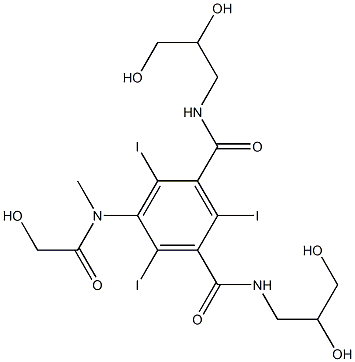
Chemical Name:Iomeprol Impurity 2
CAS.NO:104517-95-5
Synonyms:Iomeprol Impurity 2
Molecular Formula:C17H22I3N3O8
Molecular Weight:777.08529
HS Code:
Physical and Chemical Properties:
Melting point:N/A
Boiling point:N/A
Density:N/A
Index of Refraction:
PSA:
Exact Mass:
LogP:
Material Safety Information (Applicable for Hazard Chemicals)
RIDADR:
Packing Group:
Contact us for information like Iomeprol Impurity 2 chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,Iomeprol Impurity 2 physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,Iomeprol Impurity 2 Use and application,Iomeprol Impurity 2 technical grade,usp/ep/jp grade.
Related News: The complete chemical and pharmaceutical industry chain consists of basic chemical raw materials, pharmaceutical intermediates, chemical raw materials and chemical preparations. Iomeprol Impurity 2 manufacturer Higher-risk MDS is a disease with significant unmet need, and we are pleased to be able to support healthcare professionals seeking access to rigosertib, ahead of its commercial launch,�� said Mark Corbett, EVP, Inceptua Medicines Access. Iomeprol Impurity 2 supplier Higher-risk MDS is a disease with significant unmet need, and we are pleased to be able to support healthcare professionals seeking access to rigosertib, ahead of its commercial launch,�� said Mark Corbett, EVP, Inceptua Medicines Access. Iomeprol Impurity 2 vendor The New York Times said that the batches being discarded amount to around 60 million doses, citing people familiar with the matter. Iomeprol Impurity 2 factory In addition, the FDA approval ignored the recommendation of its outside advisors, who said Biogen did not provide enough evidence of clinical benefit. Three of the advisory panel’s members have resigned in protest since the FDA decision was announced on Monday.

