We serve Methyl 2-(chlorosulfonylmethyl)benzoate CAS:103342-27-4 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
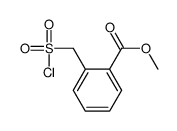
Contact us for information like Methyl 2-(chlorosulfonylmethyl)benzoate chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,o-Methoxycarbonyl benzyl sulfonyl chloride physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,Methyl 2-(chlorosulfonylmethyl)benzoate Use and application,Methyl 2-(chlorosulfonylmethyl)benzoate technical grade,usp/ep/jp grade.
Related News: Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).10-(2-Naphthyl)Anthracene-9-Boronic Acid manufacturer Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).Pentafluorobenzoic acid supplier Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).4-(methylamino)-3-nitrobenzoyl chloride vendor China is now dealing with another disease outbreak — this one mostly affecting animals but also potentially deadly among people.The quality of the drug substance determines the quality of the preparation, so its quality standards are very strict. Countries around the world have formulated strict national pharmacopoeia standards and quality control methods for their widely used drug substances.

