We serve Pentachloropyridine CAS:2176-62-7 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
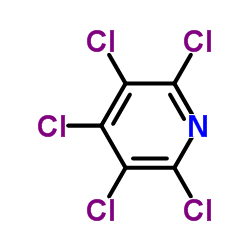
Contact us for information like Pentachloropyridine chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,3,5-dichloro-2,4,6-trichloropyridine physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,pentachloro-pyridine Use and application,Pentachloropyridine technical grade,usp/ep/jp grade.
Related News: Vietnam backtracked on Saturday and narrowed its restrictions to most flights from mainland China.Pioglitazone 2-Imine manufacturer Vietnam backtracked on Saturday and narrowed its restrictions to most flights from mainland China.Methyltrichlorosilane supplier This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?2,4-Dichloro-5-Isopropoxyaniline vendor This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?

