We serve Ramoplanin CAS:76168-82-6 to global customers since 2007, Pls send inquiry to info@nbinno.com or visit www.nbinno.com our official website should you have any interests. This site is for information only.
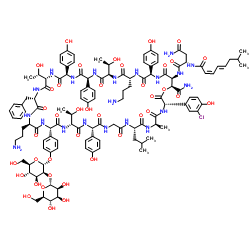
Contact us for information like Ramoplanin chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight,ramoplanin A2 physical properties,toxicity information,customs codes,safety, risk, hazard and MSDS, CAS,cas number,ramoplanin A2 Use and application,Ramoplanin technical grade,usp/ep/jp grade.
Related News: Like other countries in Southeast Asia, Indonesia depends heavily on Chinese tourism. On Thursday alone, 10,000 Chinese tourists canceled their trips to Bali, according to one industry association.1,2-Cyclohexanedicarboximide manufacturer ut others aren’t convinced — they think one of the country’s most influential state media outlets could be promoting pseudoscience and false hope.1-iodo-3,5-diphenylbenzene supplier The rigosertib Pre-approval Access Program is expected to launch in first half of 2020 and will allow Inceptua to supply intravenous rigosertib within designated countries, primarily and initially concentrated in selected countries in Europe, in response to physician requests for patients with higher-risk MDS who have exhausted all available treatment options, and are not eligible for or have no access to the INSPIRE study.Vinylmethyldimethoxysilane vendor The rigosertib Pre-approval Access Program is expected to launch in first half of 2020 and will allow Inceptua to supply intravenous rigosertib within designated countries, primarily and initially concentrated in selected countries in Europe, in response to physician requests for patients with higher-risk MDS who have exhausted all available treatment options, and are not eligible for or have no access to the INSPIRE study.Cost: Hedge prices of thick product lines + self-produced intermediates fluctuate, and sufficient supply of upstream raw materials can significantly promote the production of APIs.

